Quartz – General
What is Quartz?
- It’s a crystal found abundantly in the earth’s crust
- Chemical composition = SiO2 (Silicon Dioxide)
- When melted it becomes amorphous (non-crystalline) and is commonly known as glass, or scientifically known as fused Quartz
- You can lower its melting temperature & make it workable at lower temperatures by adding certain additives (for example, you can add soda & lime to SiO2 crystals to make soda-lime glass, which is one of the most common forms of glass used in our daily lives)
- Fused Quartz Glass is the highest-purity form of glass (no additives to lower the temperature), the general aim is to only have SiO2 with no other “impurities”
Why is purity important in Quartz Glass?
- High temperature resistance (commonly used above 1200°C), low thermal shock (withstands large and fast changes in temperature), low coefficient of thermal expansion (doesn’t grow or shrink much with large temperature variations)
- Good UV & IR transmission, used exclusively for UV (Ultra-Violet) and IR (Infra-Red) lamps where Quartz Glass is be used as a protective environment for the lamps, while still transmitting UV or IR light at high levels (80+%)
- Low reactivity to other elements (lower risk of product contamination), which makes Quartz Glass a good solution for sensitive applications where cross-contamination between the “carrier” material (in this case Quartz Glass) and the material under analysis or parts being processed must be negligible
The difference between crystalline Quartz and fused Quartz is in their microstructure. Where crystalline Quartz has a very ordered structure with regular rings, fused Quartz is less ordered. In fused Quartz the rings are of varying size, may have dangling bonds and the bonds between the corresponding ions might be strained.
The unique properties of fused Quartz are determined by either the chemical makeup (the Si-O bond) or by its microstructure:
- the wide range of thermal stability, high optical transmission and the high chemical resistance are a result of the strong Si-O bond;
- the amorphous structure is the reason for the low thermal expansion, excellent thermal shock resistance and high homogeneity of Quartz Glass
What are the key points regarding high temperature resistance of Quartz?
- Quartz sags under its own weight as a function of time and temperature
- Softening point: 1683°C, at which the glass will deform under its own weight
- Annealing point: 1215°C, at which internal stress is substantially relieved in 15 mins
- Strain point: 1120°C, at which internal stress is substantially relieved in 4 hours
- Generally below 1000°C, there is very little change in the material
- Quartz tubes sag quicker as the tube diameter is increased
- A thicker wall will extend the lifetime of a Quartz tube (wall thickness x 2 = sag/3)
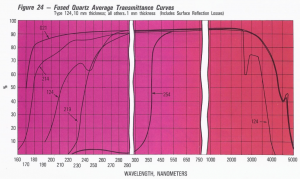
What are the key points regarding good UV and IR transmission of Quartz?
- The UV and IR transmission of Quartz Glass (ie. how much light goes through the material rather than being absorbed by the material) is directly affected by the impurity levels within the Quartz Glass
- This also means that different absorbants can be deliberately introduced to block specific wavelengths
- The transmission graph below shows average transmission levels for the main types of Quartz available on the market, where:
GE012 = synthetic Quartz, used for deep UV applications
GE214 = standard electrically-fused Quartz for tubes and rods
GE124 = standard electrically-fused Quartz for solid block material (for machining)
GE219 = Ti-doped Quartz, to block ozone level transmission
GE254 = Ce-doped Quartz, to block UV light
- Note that transmission levels may also be affected by the surface finish of the Quartz Glass.
What are the key points regarding low reactivity of Quartz?
- Quartz Glass is generally only reactive with other elements above a relatively high temperature
- One key exception to this is Na (Sodium) so it is important to ensure that Quartz is not handled with bare hands, protective gloves should be worn due to the salt content excreted from sweat glands throughout our bodies (including our hands)
- A thorough clean with alcohol (eg. isopropanol) is recommended before operation at elevated temperatures
How is Quartz produced?
- Step 1: Find a mountain with very naturally pure Quartz crystals – see weblink: BBC article on Unimin
- Step 2: Mine the Quartz rock crystals and separate them from other rocks/minerals
- Step 3: Process the rock crystals into high purity Silica sand
- Step 4: Remove contaminants as much as possible through various refining & purification methods
- Step 5: Clean & vacuum bake sand at high temperature to remove moisture
- Step 6: Melt sand in a very high-temperature and highly-controlled environment (various methods: electrical heating, induction heating, flame-fused, plasma)
- Step 7: Draw or form molten sand into finished form (most commonly tubes, rods, crucibles, or solid blocks)
- Step 8: Distribute tubes to the 1000’s of customers around the world such as (fabricators like Enterprise Q) who will transform the parts into finished products
The complexity of the process is in 1) achieving the purity of the starting SiO2 sand, and in 2) managing the hot zone of the drawing tower to control critical parameters and to ensure consistency and quality.
Quartz Glass is distinguished by its starting material. If natural raw material (e.g. Quartz sand or Quartz crystal) is used, it is called fused Quartz. If chemical precursors (e.g. SiCl4) are used, it is called fused Silica.
In addition to the raw material, Quartz Glass is distinguished by the production method. There are three established process groups that further define the properties of the Quartz Glass:
- Electrically-fused Quartz, most commonly used, lowest operating cost
- Flame-fused Quartz, traditional method, better solarisation properties and homogeneity
- Synthetic-fused Silica, exceptionally high purity, best transmission properties
Finally, for production of Quartz tubes, two primary methods are used:
- Single-step: tight tolerances, most economic, but needs a high volume to justify a run and can only run a limited range of diameters per run
- Multi-step: uses a pre-formed cylinder to re-draw into finished tubes, lower bubble content, wide availability of finished sizes, smaller batch sizes
For more information about the general properties of Quartz, further technical details, and some useful tools, please refer to the following websites: